Anna Pittermannová, Zuzana Ruberová, Juraj Petrík, František Štěpánek
Scheme of production of composite microparticles and further realease of their active cargo
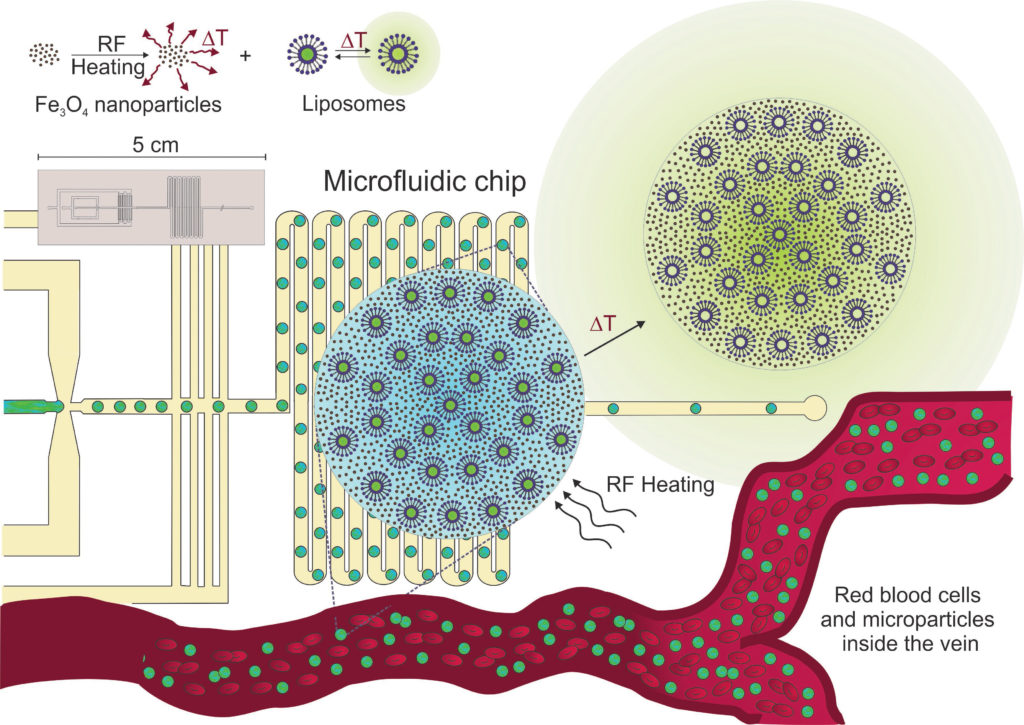
For future intravenous applications of composite microparticles in the drug delivery it is necessary to synthesize highly uniform microparticles of dimensions comparable to those of blood cells (between 5 and 10 µm). Preparation of the composite microparticles on the microfluidic chip brings us higher control over the system than other encapsulation methods and also allows us to decrease the particles size for the needed one.
Compartmentalized microparticles are synthesized from a calcium alginate gel matrix and contain several functional components, namely iron oxide nanoparticles for radiofrequency heating and MRI visualisation, liposomes for the storage and release of active molecules of their precursors. The permeability of liposomes can be significantly increased once they are heated above its phase transition temperature. The needed temperature change is achieved by iron nanoparticles upon exposure to external radio-frequency magnetic field.
Monodisperse microparticles are prepared by an extractive gelation process on a custom-designed microfluidic chip. First, droplets of aqueous solution of alginate in the continuous oil phase are formed in a flow-focusing channel. Second, gelation of the droplets is established in a long wavy channel by diffusion of calcium ions from the continuous phase. At the same time, the volume of the droplets is reduced by partial extraction of water into the continuous phase. Depending on the volumetric flow-rates of the aqueous and the oil phase, different conditions of the production are mapped and the effect of extraction on the achievable size of the microparticles is evaluated.
Nowadays we are focusing on the remotely triggered released from the microparticles as well as their surface modification for bioadhesion studies, which is a necessary step for their further applications. Another important part of our work is the design of a new microfluidic chips for the high-throughput production.
Publications
- Pittermannová A., Ruberová Z., Zadražil A., Bremond N., Bibette J., Štěpánek F., “Microfluidic fabrication of composite hydrogel microparticles in the size range of blood cells”, RSC Adv. 6, 103532-103540 (2016)
